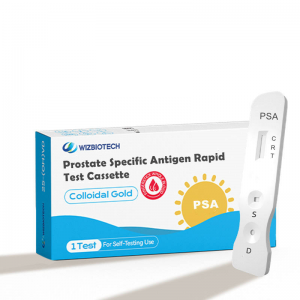
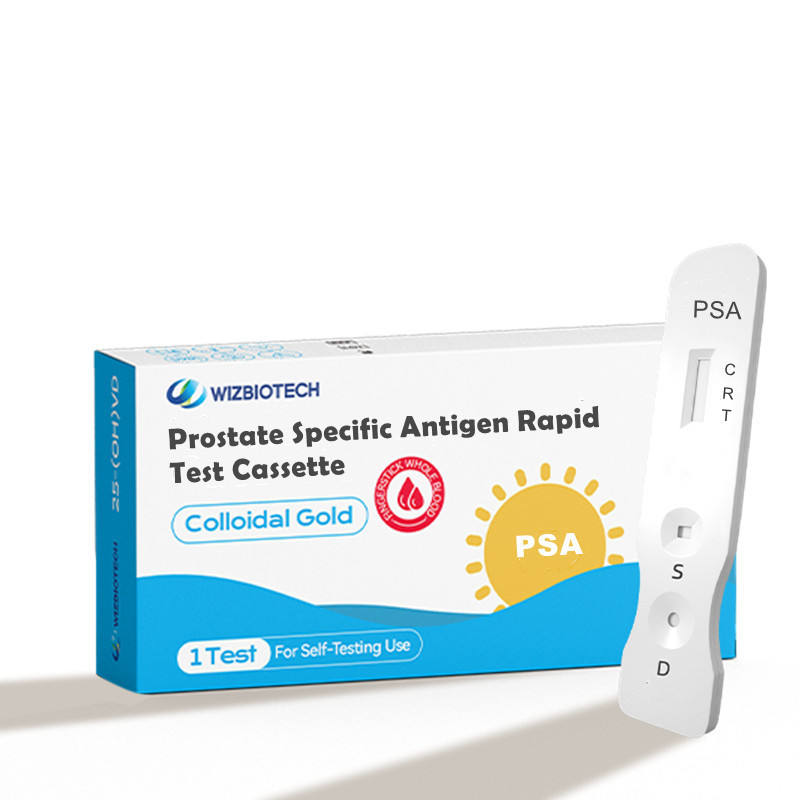
Prostate Specific Antigen PSA Self Rapid test
1. INTEDED USE
This kit is applicable to in vitro semi-quantitative detection detection of Prostate Specific Antigen inhuman serum/plasma samples and used for auxiliary diagnosis of prostatic disease. The kit onlyprovides test result of Prostate Specific Antigen. The obtained result shall be analyzed in combinationwith other clinical information. It must only be used by healthcare professionals
2. PRODUCT SPECIFICATION
| Model No. | T-PSA |
| Methodology | Colloidal Gold |
| Sample Type | serum/Plasma/whole blood /Finger Blood |
| Time to Result | 10-15mins |
| Storage | 2~30 ℃/36~86℉ |
| Shelf Life | 24 months |
| Certificate | ISO13485 |
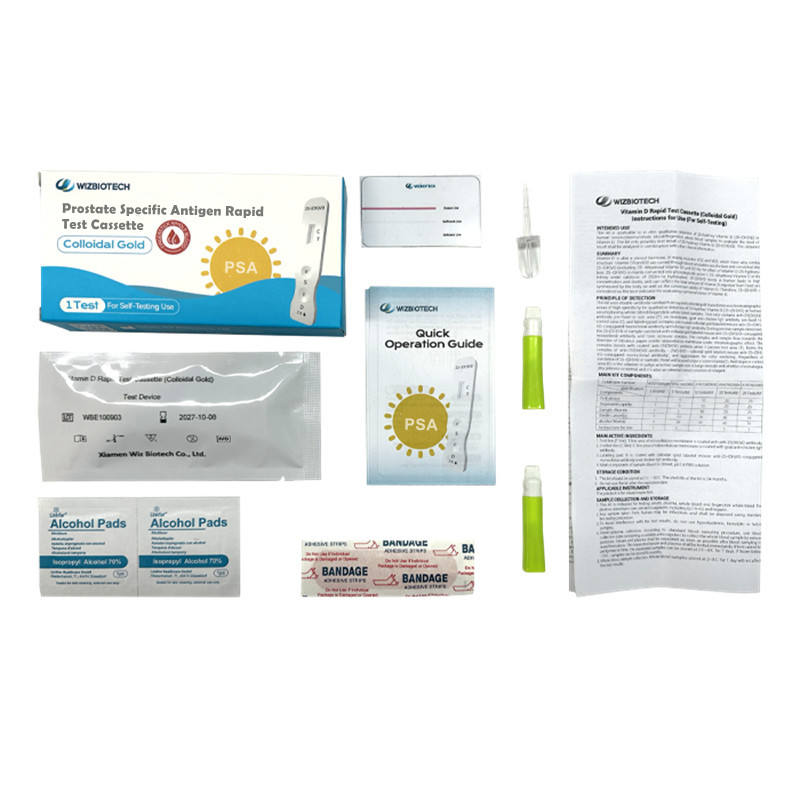
| MAIN KIT COMPONENTS
* Test device * Sample diluents * Alcohol Wipe(s) *Sterile Lancet(s) * Instructions for Use
|
PACKING
* Aluminum foil bag labeling * shrink wrap * 25 test /kit
|
3.TEST METHOD
4.OPERATION
Read the instruction for use and test operation manual completely before the test and restore the reagent to room temperature before the test. Do not perform the test without restoring the reagent to room temperature to avoid affecting the accuracy of the test results.
1.Remove test device from aluminum foil pouch, lie it on a horizontal workbench, and do a good job in sample marking.
2.Gently touch the pipette tip to the blood drop(serum/plasma/whole blood/fingerstick whole blood). The pipette will self-fill with the required volume (10 μL).
3.Release blood sample by touch the tip of disposable pipette to sample pad.
4.Release the blood sample onto the "S" well sampling pad by touching the tip of the disposable pipette..
5.Remove the cap of sample diluent. Vertically add 3 drops to the D hole.
6. Interpret result within 10-15 minutes, and detection result is invalid after 15 minutes (see detailed results in result interpretation)
5. CERTIFICATE
* ISO System Certificate
* CE Certificate
* EU Registration
* UCKA MHRA Registration
6.EXHIBITION

7.GLOBAL PARTNER